Public advisory - Unauthorized and counterfeit health products seized at Thumbs Up 4 Shiatsu in Mississauga may pose serious health risks Français
OTTAWA, ON, March 25, 2026 /CNW/ -
Summary






- Product: Unauthorized and counterfeit health products, including prescription and injectable drugs.
- Issue: Health products – Unauthorized and counterfeit products; Product safety
- What to do: Do not use these products. Consult a licensed healthcare professional if you have been administered or have used these products and have health concerns.
Affected products
Unauthorized and counterfeit health products seized from Thumbs Up 4 Shiatsu (located at 3465 Semenyk Court, 2nd floor, Mississauga, Ontario), including:
Category |
Product name |
Unauthorized health products containing prescription drugs |
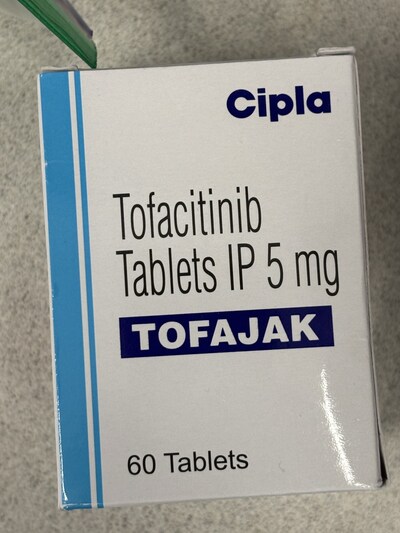
Tofacitinib Tablets IP 5 mg |
Rhofanib 5 (Tofacitinib 5 mg) |
|
Haircare, Laboratoire Revitacare France (Hyaluronic acid 2 mg) |
|
Unauthorized injectable drugs |
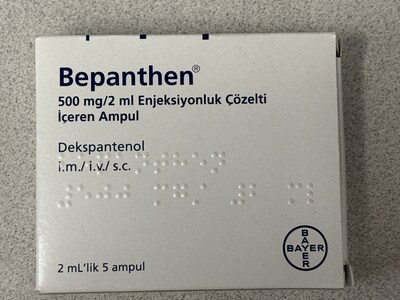
Bepanthen (Dekspantenol 500 mg/2 ml) |
Bepacto (Dexpanthenol 500 mg/2 ml)) |
|
Biacto (Biotin 5 mg/1 ml) |
|
Counterfeit health products |
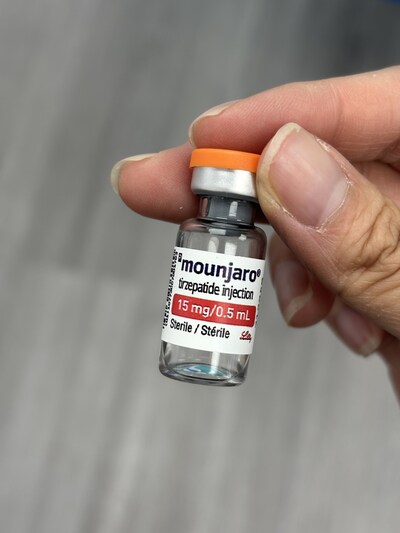
Mounjaro vials (tirzepatide) |
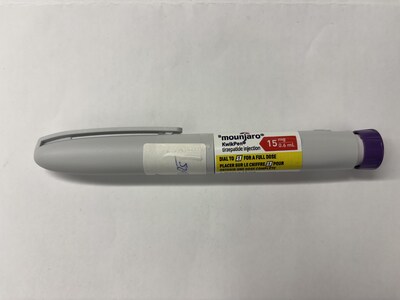
Mounjaro KwikPen (tirzepatide injection) |
|
KwikPen (tirzepatide injection), Drug Identification Number (DIN) 03552000 and batch D856216 are invalid |
Issue
Health Canada is warning consumers about multiple unauthorized and counterfeit health products seized at Thumbs Up 4 Shiatsu in Mississauga, Ontario. Health Canada has not authorized any of the health products that were seized, which means that they have not been assessed for safety, efficacy and quality. Selling unauthorized or counterfeit health products is illegal.
The seized products, including injectables are labelled to contain prescription or other drug ingredients. These products may have been sold or administered for pain management, hair loss, weight management or other cosmetic purposes.
Counterfeit drugs are made to look like authentic products, but they are not the same and they may not contain the drug at all. Though some of the counterfeit products are labelled with DINs, which may lead consumers to believe the products were authorized, the manufacturer of Mounjaro has confirmed that the seized products are counterfeit. The best way to make sure your prescription drugs are authentic is to buy them from a licensed pharmacy with a prescription from a healthcare professional. Prescription drugs cannot be sold outside of a licensed pharmacy and should only be taken under the care of a healthcare professional because they are used to treat specific health conditions and may pose serious health risks.
Unauthorized or counterfeit health products may:
- Interact with other medications an individual might be taking.
- Contain too much, too little, or none of the active ingredient.
- Contain high-risk ingredients, additives, or contaminants that may or may not be listed on the label.
- Have not been manufactured or stored safely.
Unauthorized or counterfeit injectable drugs carry additional risks, including infections, scarring, allergic reactions, and other serious adverse outcomes due to contamination, improper handling, or unsafe administration.
Should additional safety concerns be identified, Health Canada will take appropriate action to protect public health and safety.
What you should do
- Do not buy or use unauthorized or counterfeit health products. When receiving cosmetic treatments, read product labels (or ask your healthcare professional to show you the product label) to confirm a product has been authorized for sale by Health Canada. Authorized health products have an eight-digit DIN, Natural Product Number (NPN) or Homeopathic Drug Number (DIN-HM).
- You can also check whether products have been authorized for sale by searching Health Canada's Drug Product Database, Licensed Natural Health Product Database or the Medical Devices Active Licences Listing (MDALL).
- Only buy prescription drugs from licensed pharmacies.
- Consult a licensed healthcare professional (physician, nurse practitioner, pharmacist) if you have been administered or have used unauthorized health products and have health concerns.
- Report any health product-related side effects or complaints to Health Canada.
- Learn more about buying health products safely:
Background
Tirzepatide is available only by prescription in Canada for the treatment of adults with type 2 diabetes (under the brand name Mounjaro) or for chronic-weight management (under the brand name Zepbound). Tirzepatide can be associated with significant risks, which include gastrointestinal side effects (nausea, vomiting and diarrhea), pancreatitis, worsening kidney function, worsening depression or suicidal thoughts, hypoglycemia (low blood sugar when combined with other diabetes drugs), potential thyroid tumours, diabetic retinopathy (eye) complications, hypersensitivity reactions (i.e., allergic responses, including anaphylactic reactions), and delayed gastric emptying (i.e., the stomach taking too long to empty food into the small intestine) that may pose risks if the patient is undergoing a procedure which requires sedation. Tirzepatide should not be taken by pregnant or breastfeeding women.
Tofacitinib, also known under the brand name Rhofanib, is a prescription medication that affects the immune system and that is only approved to treat specific medical conditions (rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis ulcerative colitis and juvenile idiopathic arthritis) at recommended doses. Taking tofacitinib for other uses without guidance from a licensed healthcare professional can pose serious health risks. The medication suppresses parts of the immune system and may increase the risk of infections, blood clots, cardiovascular events, and certain cancers, particularly if taken without proper medical oversight. The risks may be greater for certain patient populations, including older adults, individuals with a history of cardiovascular disease, smokers, or those with weakened immune systems or chronic infections. Patients should be supervised by a qualified healthcare professional with appropriate screening and follow-up to track side effects and to prevent complications.
SOURCE Health Canada (HC)

Media Enquiries: Health Canada, (613) 957-2983, [email protected]; Public Enquiries: (613) 957-2991, 866 225-0709, [email protected]
Share this article