Prometic Reports Positive Clinical Data From Ongoing PBI-4050 Study in Alström Syndrome Patients Français
- Clinical benefits in both heart and liver observed in patients treated with PBI-4050 over an average of 52 weeks of treatment
- Cardiac MRI indicates PBI-4050 reversed trend of fibrosis progression
- Reduction in liver fibrosis in patients treated with PBI-4050 demonstrated by MRI and Fibroscan
- Upcoming meetings with FDA and EMA to define PBI-4050's clinical-regulatory pathway in Alström syndrome
LAVAL, QC, March 28, 2018 /CNW Telbec/ - Prometic Life Sciences Inc. (TSX: PLI) (OTCQX: PFSCF) (Prometic) today announced new clinical data from its ongoing Phase 2 open label clinical trial being conducted in the United Kingdom investigating PBI-4050 for the treatment of patients with Alström syndrome.
The clinical study, which has enrolled 12 patients, reported that clinical activity and tolerability of PBI-4050 are sustained with prolonged treatment. The average treatment duration of PBI-4050 for the 12 patients has reached 52 weeks and further clinical activity in the heart and liver was observed with longer treatment exposure.
"Progressive cardiac fibrosis is the most serious feature of Alström syndrome, and the resultant heart failure is the most common cause of death," said Dr. John Moran, Chief Medical Officer of Prometic. "Therefore, the results observed with the cardiac MRI are very promising and exceeded our expectations. Going into the study, we had been hoping to see a slowing of the rate of progression, but in fact, we have observed a regression in cardiac fibrosis. Additionally, the liver MRI further supports the FibroScan scores and the reduction of fibrosis observed in these patients. Importantly, PBI-4050's clinical activity and tolerability have been confirmed over this extended period, with no drug-related serious adverse events. The clinical activity observed in these patients gives us further confidence in the forthcoming pivotal Phase 3 clinical trial in patients with idiopathic pulmonary fibrosis."
Pierre Laurin, President and Chief Executive Officer of Prometic, stated, "We are scheduled to meet this summer with the European and the U.S. regulatory authorities to determine the clinical-regulatory pathway for this investigational treatment for patients with Alström, who are in serious need of an anti-fibrotic treatment. This is a critical step to determine whether this ultra-rare pediatric disorder could be an indication granted priority review."
Summary of Cardiac Data
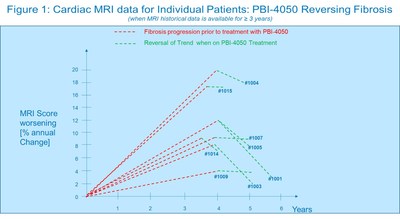
Analysis of the interim cardiac MRI data indicates a reduction of cardiac fibrosis in each patient after initiation of treatment with PBI-4050 (p<0.001). Figure 1 below illustrates the progression of cardiac fibrosis expressed as a percent increase of the MRI score for each patient for whom three years or more of fibrosis data were available, and the reversal of said progression when patients were treated with PBI-4050. The length of the red dashed lines corresponds to the durationof fibrosis data and the length of the green dashed lines to the duration of PBI-4050 treatment for each patient.
Summary of Liver Data
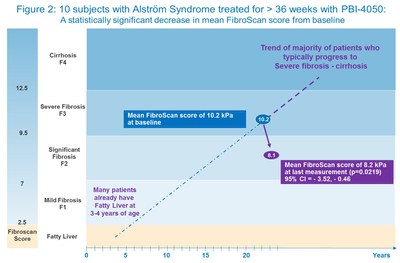
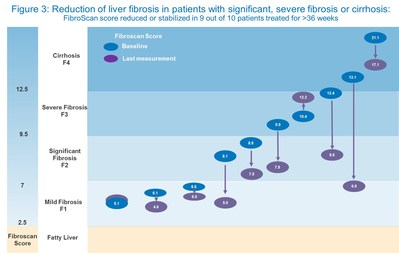
Fibroscan results from the 10 subjects who received at least 36 weeks of treatment showed a statistically significant improvement in the measure of liver stiffness, from a mean of 10.2 kPa at baseline to a mean of 8.1 kPa at last measurement, an absolute decrease of 2 kPa (p = 0.0219, 95% CI -3.52, -0.46) (Figures 2 & 3). Fibroscan is a non-invasive technique for clinical assessment of liver fibrosis with a high degree of accuracy and reproducibility, especially in patients with established fibrosis (≥ F2) (Cassinotto 2016).
Liver MRI data also indicated a mean reduction of -11% in the T1-corrected score between baseline and last available measurement (p=0.0195, 95% CI: -92.3, -9.8), which supports an improvement of liver fibrosis.
Positive effects on other parameters of the liver have also been observed and will be disclosed at The International Liver Congress™ 2018, the annual meeting of the European Association for the Study of the Liver (EASL), to be held in Paris, April 11-15, 2018.
More about Alström syndrome:
Alström syndrome is a rare inherited autosomal recessive syndrome characterized by the onset of obesity in childhood or adolescence, Type 2 diabetes, often with severe insulin resistance, dyslipidemia, hypertension and severe multi-organ fibrosis involving the liver, kidney and heart. Alström syndrome is also characterized by a progressive loss of vision and hearing, a form of heart disease that weakens the heart muscle (dilated cardiomyopathy), and short stature. This disorder can also cause serious or life-threatening medical problems involving the liver, kidneys, bladder, and lungs. The clinical manifestations of Alström syndrome vary in severity, and not all affected individuals have all of the features associated with the disorder.
More about PBI-4050
PBI-4050 is an orally active lead drug candidate with excellent safety and efficacy profiles demonstrated in a large number of animal models of fibrosis affecting different organs, including the lung, liver, heart, kidney, and pancreas. The effects of PBI-4050 demonstrated in animal models have been replicated in Phase 2 studies in IPF, in metabolic syndrome with type 2 diabetes and in Alström syndrome. PBI-4050 is entering pivotal placebo-controlled phase 3 clinical trials for the treatment of IPF and has already started placebo-controlled phase 2 trials in metabolic syndrome and type 2 diabetes patients.
More about Prometic
Prometic Life Sciences Inc. (www.prometic.com) is a biopharmaceutical corporation with two drug discovery platforms focusing on unmet medical needs in the field of fibrosis and orphan diseases. The first platform, small molecule therapeutics, stems from the discovery of two receptors GPR40/GPR84 acting as "dual master switches" which are at the core of the healing process as opposed to fibrosis. The second platform, plasma-derived therapeutics, leverages Prometic's vast experience in bioseparation technologies to address unmet medical needs with therapeutic proteins not currently commercially available, such as Ryplazim™ (plasminogen human). Prometic is also leveraging the second platform higher recovery yield advantage to develop some more established plasma-derived therapeutics with significant growth in demand such as Intravenous Immunoglobulin (IVIG) and provides access to its proprietary bioseparation technologies to enable pharmaceutical companies in their production of non-competing biopharmaceuticals. Globally recognized as a bioseparations expert, the Corporation derives revenue from this activity through sales of affinity chromatography media which contributes to offset the costs of its own R&D investments. Headquartered in Laval (Canada), Prometic has R&D facilities in the UK, the U.S. and Canada, manufacturing facilities in the UK and commercial activities in the U.S., Canada, Europe and Asia.
Forward Looking Statements
This press release contains forward-looking statements about Prometic's objectives, strategies and businesses that involve risks and uncertainties. These statements are "forward-looking" because they are based on our current expectations about the markets we operate in and on various estimates and assumptions. Actual events or results may differ materially from those anticipated in these forward-looking statements if known or unknown risks affect our business, or if our estimates or assumptions turn out to be inaccurate. Such risks and assumptions include, but are not limited to, Prometic's ability to develop, manufacture, and successfully commercialize value-added pharmaceutical products, the availability of funds and resources to pursue R&D projects, the successful and timely completion of clinical studies, the ability of Prometic to take advantage of business opportunities in the pharmaceutical industry, uncertainties related to the regulatory process and general changes in economic conditions. You will find a more detailed assessment of the risks that could cause actual events or results to materially differ from our current expectations in Prometic's Annual Information Form for the year ended December 31, 2016, under the heading "Risk and Uncertainties related to Prometic's business". As a result, we cannot guarantee that any forward-looking statement will materialize. We assume no obligation to update any forward-looking statement even if new information becomes available, as a result of future events or for any other reason, unless required by applicable securities laws and regulations. All amounts are in Canadian dollars unless indicated otherwise.
SOURCE ProMetic Life Sciences Inc.

Corporate Contacts: Pierre Laurin, President and CEO, Prometic Life Sciences Inc., [email protected], 450.781.0115; Frederic Dumais, Senior Director, Communications & Investor Relations, Prometic Life Sciences Inc., [email protected], 450-781-0115; Investor Contact: Bob Yedid, LifeSci Advisors, [email protected], 646-597-6989; Media Contact: Matt Middleman, M.D., LifeSci Public Relations, [email protected], 646-627-8384

Share this article